The Technology House now offers AMT's PostPro3D patented vapor smoothing technology to help reduce time in post processing and provide quality parts with a higher level finish.

The BLAST process is a physical-chemical-based process that can smooth a wide variety of thermoplastic polymers. The process is a non-line-of-sight and can smooth complex internal cavities of polymer parts. The PostPro3D technology is highly controllable, allowing reproducible results with no degradation of a parts’ mechanical properties. Using a series of pre-defined parameter sets and algorithms PostPro3D achieves for the first time a surface finish that matches injection molding techniques. This
enabling technology reduces lead-time, cost of manufacture, operational and maintenance costs providing the ‘missing piece’ in the digital manufacturing chain. Here is a list of all the benefits it offers.
The added capabilities keep TTH's customers at the forefront of their field. The Technology House can now integrate the AMT PostPro3D into its digital workflow to continue to provide customers with an end to end solution for production. Among the improvement in surface finish qualities, the AMT PostPro3D offers a wide range of benefits listed below.
WIDE RANGE OF MATERIALS
AMT is printer and material agnostic which allows for full digital integration in the manufacturing workflow. Below is a quick snap shot of compatible printers and materials. You can download the full list here.
Printer and Material Capability
Powder Bed Fusion SLS | MJF | HSS
Extrusion FFF | FGF | HSE
Rigid Plastics PA6 | PA11 | PA12 | ABS | PC | Polypropylene
Elastomers TPU | TPE | SBC | PEBA
Composites Glass & Carbon-filled
REDUCE PART COST
IMPOVED SURFACE ROUGHNESS
The PostPro3D technology smooths the surface of a polymer part using a program with a selection of a predefined set of parameters. Each set of samples was processed in separate batch using a different
parameter set. Each complete process run took 1 hour.
The surface roughness of the samples was measured using Mitutoyo Surftest SJ-210 surface roughness measurement device with the stylus tip radius of 2μm, tip angle 60° and measuring force 0.75kN. Five measurements at different areas of each surface were made before and after processing and the average taken. This was performed in accordance with ASTM standard ASTM D7127. The three standard surface finishes are compared to un-processed (as-printed) surfaces as shown below. Note the repeatability of the smoothed surfaces is demonstrated by the small standard deviation.

MECHANICAL PROPERTIES
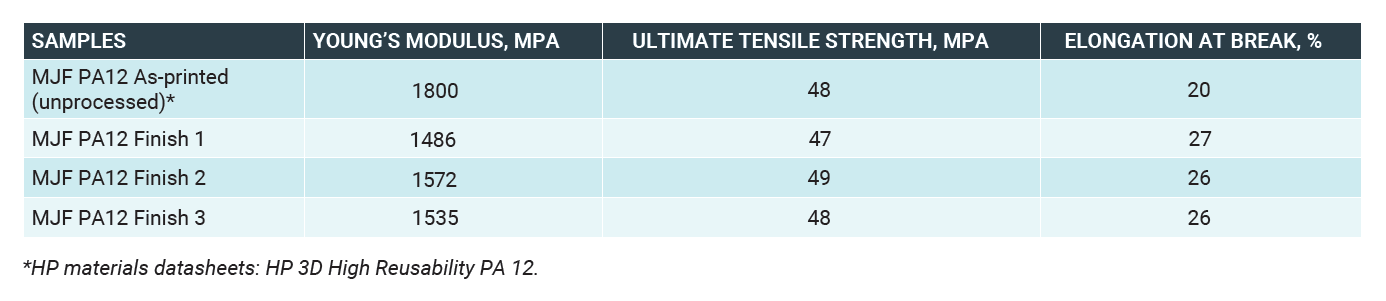
Ultimate tensile strength, elongation at break and Young’s modulus were measured for the processed and un-processed PA 12 samples. Samples were prepared to ASTM D638 Type 1 dimensions. The gauge length of the tensile testing machine was 50mm and tested at a speed of 5mm/min.
The results show no loss in Ultimate Tensile Strength in processed samples. Elongation at Break (EAB) of the tested samples has significantly increased, whereas the Young’s Modulus decreased. The increase in EAB can be explaining by the reduction of crack initiation sites on the polymer surface due to the removal of surface porosity, as shown in the microscopy image below. The results are summarized in the table below:


WATER ABSORPTION
3D printed parts are porous and rough. AMT cleans, smooths and seals the surface. This enables the ability to pass regulatory testing for
end-use in various industries.
CYTOTOXICITY
The cytotoxicity test is one of the biological evaluations and screening tests that use tissue cells in vitro to observe the cell growth, reproduction and morphological effects by medical devices. Cytotoxicity is an important indicator for toxicity evaluation of medical devices as it is simple, fast and has a high sensitivity.
Cytotoxicty tests were carried out at a nationally recognized European laboratory to Normative References: ISO 10993-5 (2009); ISO 10993-1 (2010); ISO 10993-12 (2012).
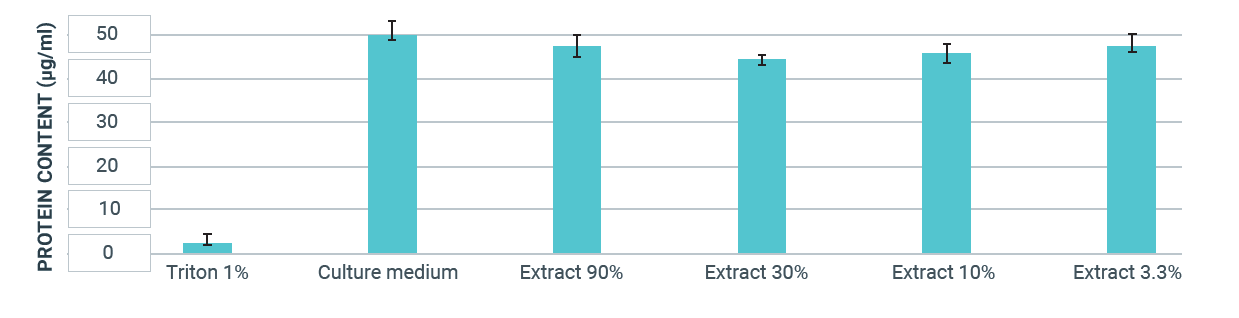
The results show that in the presence of Triton X 100 in the cell culture medium, 6.0 % of the protein content compared to the negative control was reached. This value is within the valid range of 15 % protein content or less compared to the negative control. Materials are considered cytotoxic, if the material extract leads to a protein content of the test cells of less than 70 % compared to the negative control.
This was not the case in this test. The material extract therefore does not show a cytotoxic effect.
Ready to see how Post Pro3D can unlock digital production for your project? Contact us to get started.







.jpg)

.png)

.png)

